
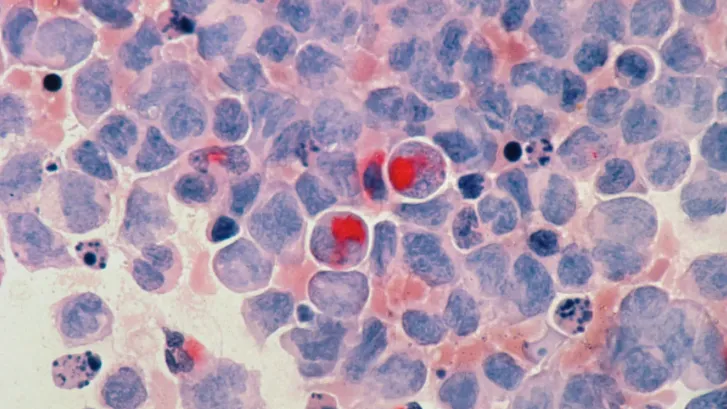
Underfunding hampers cancer care in Asia: health economist
Survival rates are lower in middle-income Asian nations despite lower cancer incidence.
Cancer care in Asia has been hampered by underfunding of its health care systems and the absence of universal health coverage, putting patients at risk of “financial catastrophe,” a health economist has said in a blog post published by the Asian Development Bank (ADB).
Thomas Hofmarcher of the Swedish Institute for Health Economics said the annual number of new cancer cases increased from 4.9 million in 2002 to 9.5 million in 2020, and this is expected to worsen due to an unprecedented rate of population ageing.
Although high-income countries with higher proportions of older populations, such as Japan or the Republic of Korea, face much higher cancer numbers than middle-income countries, this pattern is reversed in survival rates, he said.
READ MORE: South Korea urged to revise cancer tests reimbursement system
In the wealthier countries of Asia and the Pacific region, such as Australia, Japan, and the Republic of Korea, around 56–65 in every 100 new patients survive cancer, whereas in the developing part of the region only around 28–40 in every 100 new patients survive.
“In Asia, early detection is hampered by low awareness of the early signs of cancer among the general population and the absence of or low participation in screening programs, such as for breast cancer,” Hofmarcher said.
In addition, cancer patients in lower-income Asian countries incur full out-of-pocket costs for some generic medicines, which are readily available in higher-income nations, because several of the most basic cancer medicines listed on the World Health Organization’s Model List of Essential Medicines are not listed on their national formularies.
Hofmarcher blamed this on underfunded healthcare systems and the absence of universal health coverage, especially in India, Indonesia, the Philippines, and Vietnam.
“As many as half of all cancer patients and their households in Southeast Asian countries face financial catastrophe from high out-of-pocket medical bills and related expenditures in their first year after cancer diagnosis,” he said.
To address these problems, Hofmarcher recommended a “value-based and evidence-based” inclusion of new medicines in national formularies, the use of potential savings by governments in the region to stimulate competition among pharmaceutical producers, and stricter price control of originator medicines after patent expiry to create budget headroom for governments.
“These savings could be re-channelled to fund and list innovative medicines, offering a more sustainable financing model whilst improving patient outcomes,” he said.











 Advertise
Advertise








